The aim of the NIHR BRC Digital Therapies theme is to help bridge shortfalls and close gaps in NHS service provisions with digital solutions.
To achieve this, we need acceleration in the development and implementation of evidence-based, novel digital therapies. One major challenge is that the applicable UK regulation of Software and Artificial Intelligence as medical device, is still evolving under the Medicines and Healthcare products Regulatory Agency (MHRA) Software and Artificial Intelligence as a Medical Device Change Programme. The regulation complex to navigate, and impacts every stage of the digital therapy lifecycle. The UK government’s currently proposed, new Medical Technology (MedTech) pathway outlines the ‘rules’ and principles on which NICE and NHS England will work in partnership to evaluate, commission and fund medical technologies.
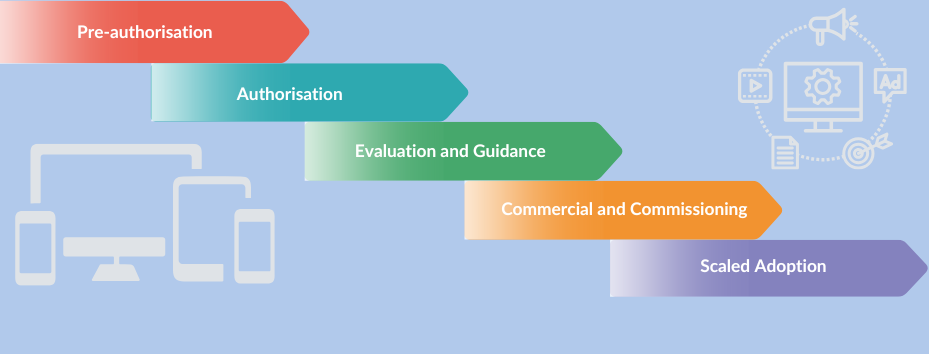
Knowing what is required from creators of digital therapies in each of the five phases of the MedTech pathway (Pre-authorisation, Authorisation, Evaluation and Guidance, Commercial and Commissioning, Scaled Adoption) is essential to avoid costly and time-consuming mistakes with serious and potentially legal implications. To overcome this challenge, we have developed these web pages to help people understand and navigate the UK MedTech pathway specifically for digital mental health therapies, a sub-set of Software and Artificial Intelligence as medical device.