The UK has a nationalised healthcare system with four National Health Services for each of the devolved nations. National health services are overseen by the UK government’s Department of Health and Social Care (DHSC) who sets UK health and social care policy and transfers funding to NHS England. In this guide we will focus on processes affecting NHS England.
NHS England (NHSE) has a complex organisational structure. There are 42 Integrated Care Systems (ICSs), who ‘bring together their local health and care organisations to improve outcomes, tackle inequalities and create better services’. NHS England passes the majority of funding on to ICS boards for budget administration that is responsive and tailored to local needs. Therefore the commissioner and payor for medical devices, specifically for digital mental health therapies, will generally be one of the 42 ICS boards.
Practical implementation of commissioned services, which includes medical devices and therefore digital therapies, depends on local NHS service infrastructure which consists of trusts which vary widely by service type and geographic area.
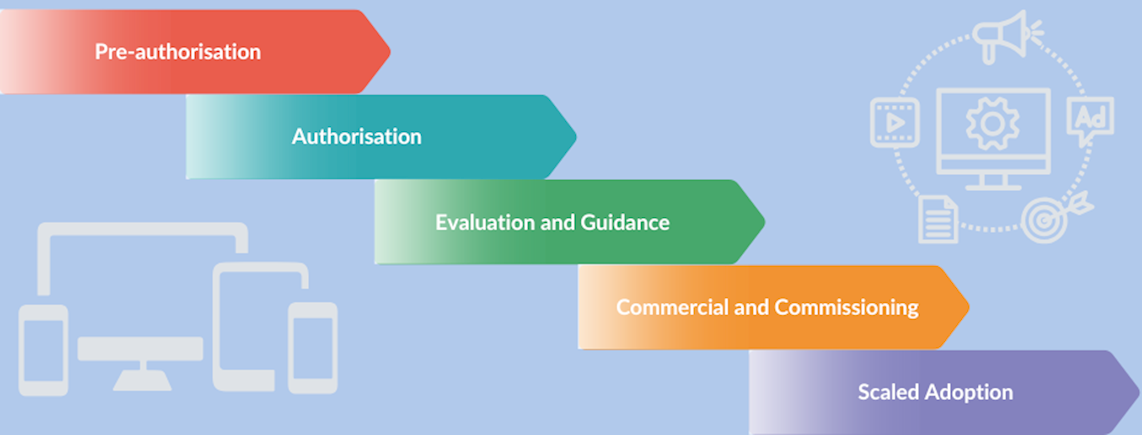
The proposed MedTech pathway
Conditions for commissioning
Health technologies, which include medical devices such as digital therapies, can only be commissioned under two conditions. Firstly, health technologies must meet UK regulatory standards, which are assessed by the Medicines and Healthcare products Regulatory Agency (MHRA). Secondly, health technologies must meet clinical and cost-effectiveness requirements, which are assessed by the National Institute of Health and care Excellence (NICE) in England, Health Technology Wales, the Scottish Health Technologies Group (SHTG), and the Department of Health in Northern Ireland. NICE provides an evidence standards framework for digital health technologies with advice on how to meet each.
The UK government’s Department of Health and Social Care published its inaugural medical technology (MedTech) strategy policy document in February 2023; and its first annual update 09/April/2024. Within this the proposed MedTech pathway sets out the steps required to pass the commissioning stage and progress to NHS adoption.
Pilot scheme for innovative devices
This is currently being piloted under the Innovative Devices Access Pathway (IDAP) pilot scheme, Eight diverse innovative health technologies were chosen for the IDAP pilot, including one digital therapy: A fatigue management app for people with multiple sclerosis, REFUEL-MS. The REFUEL-MS app implements NICE-recommended symptom treatment guidelines, and is funded by a grant awarded by the National Institute for Health and Care Research (NIHR) and co-funded by the Multiple Sclerosis Society (award ID: NIHR203290). It is developed by a collaboration between an academic and commercial technology partner: King’s College London and Avegen Ltd and the chief investigator is the NIHR Maudsley BRC’s digital therapies theme lead Rona Moss-Morris.

